Clinical Trial Management System (CTMS)

Automate Clinical Trial Operations & Efficiently manage clinical data for your trials and registries
VST research’ Clinical Trial Management System (CTMS) enables researchers to easily capture and integrate data from clinicians, patients, devices, wearables, and EHR systems.
CTMS is designed to provide quick and easy access to study or subject information utilizing simple and intuitive navigation. In most cases, needed information is only a few clicks away. CTMS will guide you from study start-up to study close-out, and everything in between.
Securely track and manage study activities
The highly configurable role-based access and permissions in CTMS make it easy for research teams to perform tasks in a regulatory compliant, secure manner. Robust reporting and analytics capabilities allow you to closely follow study progress and performance, ensuring important milestones are met at every level.
Site Management
Keep your sites productive and compliant.
Give sites an easy, and intuitive way to provide accurate study data. Auto-notify site users with actions required.
- Check out site to laptop for monitoring manually or via email
- Complete electronic regulatory monitoring forms and subject visit/ CRF monitoring requirements at site
- Track issues and required actions for monitored items
- Capture notes for monitored items
- Check in monitoring issues/results once visit is complete
- Auto-manage study calendars.
- Apply electronic signatures.
Data Managers
Maintain control over your study data.
Build, preview, publish, and repeat on a collaborative, drag-and-drop interface. Provision sites and users, resolve queries, and export datasets in a flash.
Monitors
Simplify your monitors work.
Give your monitors a mobile friendly dashboard to assist them in the field, showing what they need to do, and when they need to do it. Review site activities, verify data against source.
Participants
Get frictionless data capture from patients.
Allow patients to provide data anytime, anywhere from their own device. Provide timely notifications when data is required.
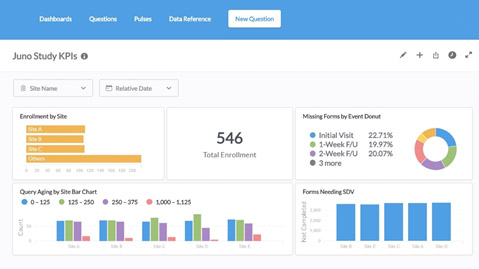
Sponsors
Keep up-to-date with study progress. Save time and money. Maintain regulatory compliance. Rest well knowing your data is secure.
IRB/EC Management
- Track Site IRB/EC submissions
- Track multiple protocol submissions per study
- Use alerts to identify expiring IRB approvals
- Track approval and reapproval of protocol and informed consent
Randomization
Clinical trial randomization can be complex. Assigning subjects and managing supplies shouldn’t be.
- Easy to use for all types of randomized clinical trials (RCTs)
- Randomize from within CTMS system
- Simple, blocks, random permuted blocks, minimization
- Stratify by age, sex, site, or any CRF variable
- Unlimited treatment groups, custom allocation ratios
- Generate randomization lists
- Email notifications
- Supports blinded studies
- Allocate randomization codes at follow-up visits
- Code list management
- Generate randomization codes for labeling treatment kits
- Integrate randomization into your study workflow
Patient Database Management
- CTMS’s easy navigation allows all users to run searches within the system based on multiple criterion: Age, Gender, BMI, Inclusive/Exclusive Medical Conditions, Inclusive/Exclusive Medications.
- At the click of a button, send an email to the subjects matching your search criterion
- Makes filling out feasibility questionnaires easy
- Export lists to Excel with the click of a button
Accounting and Budgeting
- Build your budget templates according to the study event schedule.
- Create invoices to sponsors for completed/ monitored visits or other study revenue items as needed.
Payments Manager
Delayed payments continue to be a source of site dissatisfaction with the clinical trial process. In the CTMS, the payment process is automatically triggered when sites complete tasks such as data entry in CTMS. And, both sites and sponsors have the same access to the payment history in system.
- Track completed forms and visits for payment purposes
- Assign specific monetary amounts to individual forms across all study visits
- Manage and track payments, as needed
- Set up user-defined payment rules and conditions
- View the transactional log of all previous payments
- Integrate with third-party payment processors
Study Documents/File Management
- Manage documents via a centralized study document manager
- Upload documents to any CRF
- Access documents via the document viewer
- Annotate documents by user
- Redact documents by user
Site Regulatory Documents
- Manage documents via a centralized site regulatory document manager
- Define site enrollments based on site document completion rules
- Create forms to assign and track regulatory documents by site
- Link enrollment to regulatory document completion
- Show site-based status of regulatory document completion
Study-related Forms
- Capture study-related data such as the protocol or study materials/inventory
- Build and deploy study-related forms such as protocol approvals, financial disclosures, delegation of authority, regulatory log, and site essential docs to the web
Study Calendar
- Easily schedule study specific visits. View calendars based on individuals, teams, investigators, or the entire company.
- Invite additional team members to your study appointments.
- Reminders and Notifications
- Automatically send appointment confirmations and reminders to all your patients
Analysis & Reporting
Insight is the intelligent way to visualize, analyze, and share your study data.
- Create your own prescreening logs so you can stop filling them out by hand.
- Compare financial data by coordinator, investigator or entire company.
- Ensure patient recruitment is on time and within budget with multiple reports specifically for recruitment.
- Track advertising budgets and effectiveness of your campaigns.
- Financial study reports show study earning costs and profitability.
- Export in CSV, Excel, SPSS or R
- Analyze in the tool of your choice (SAS, STATA, SPSS or R)

Compliance Management
The CTMS Compliance Support Service makes it easy for our customers to achieve, maintain, and demonstrate compliance with:
- Regulations (e.g. from FDA, EMA, including 21 CFR Part 11, HIPAA, GDPR, ICH-GCP),
- Data practices and security standards (ISO, SOC), and
- Institutional policies (e.g. your SOPs, security assessments, etc.).
The service provides you with continuously maintained documentary evidence, access to CTMS’s internal Quality System, and ongoing support and guidance for compliance-related issues.
Support and Training
- Quick and efficient user training
- In-person training also available onsite at sponsor facilities
- Zero client requirements except browser
- Remote training available
- Technical support and dedicated support number per study
- Many levels of help desk and support options available
Security
- Role and workflow-based security
- Customizable event notifications
- Configurable workflow to fit customer’s organization
- Configurable down to the field level
- Configurable actions by role or user
- Enterprise class data encryption and user log-in security.
Data / Hosting
- All data will be hosted on a validated, secure server located in a hardened, redundant hosting facility
- Professional data centers ensure that your data is secure.
- HIPAA compliant subject information storage.
- Full business continuity and disaster recovery functions provided
- All data will be backed up on a nightly basis both locally and to a different geographic location for redundancy
- 24/7/365 Performance and fault monitoring
- All files will be the client’s property and transferred upon request
For more information Contact VST Research.

